Scalable eQMS software
to make quality easy
Trusted quality software to help growing
life science companies in the US 🇺🇸 get to market faster
Request a demo

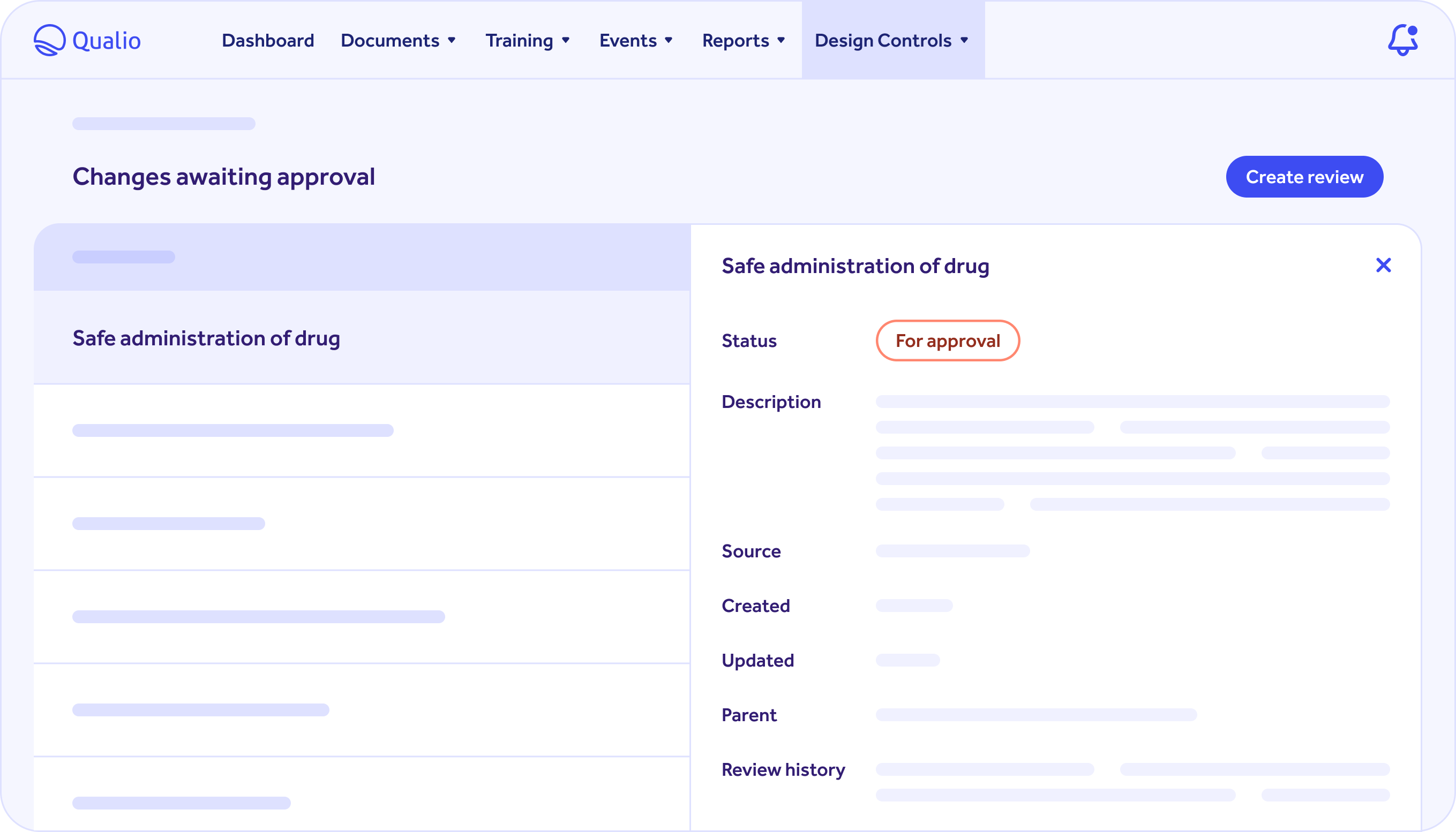
THE QUALIO PLATFORM
Qualio offers a scalable system for your team that is easy to implement and use
Our platform scales across teams and products as your company grows and, unlike paper or spreadsheets, is completely flexible.
Qualio is rated as the #1 “easiest setup” and “easiest to use” of all eQMS providers reviewed on G2. Our expert team ensures a quick implementation and our intuitive platform is loved by everyone from quality experts to basic users.
OUR SOFTWARE
Qualio is purpose-built for life sciences
We provide targeted software for the entire life science ecosystem.
Find out how we are the right fit for your company.
Medical Devices
Streamline a fast, safe pathway to market for your medical device while ensuring your teams stay compliant.
Pharmaceutical
Embed industry-leading pharmaceutical quality processes that simplify and unlock FDA, ISO and ICH compliance.
Biotech
Beat the competition with quality-centric products that satisfy regulators, patients and stakeholders.
Cannabis
Earn regulatory trust and position your business for growth with a robust and quality-first approach.
Software as Medical Device
Unlock end-to-end control of your entire product lifecycle, connecting teams and cutting stress.
Contract Research Orgs
Deliver a compliant and continuously improving CRO service that delights clients and brings new ones flocking.
QUALIO INTEGRATIONS
We do the heavy lifting so you can focus on what matters most
Qualio integrates with your business-critical applications to let you automate cross-functional workflows, share data and build a single source of truth for all your quality information.